Invotec is now offering a new equipment validation program to support medical device manufacturers facing implications from the new European Medical Device Regulation (EU-MDR).
Set to take effect this year, the EU-MDR will require medical device manufacturers to meet higher documentation requirements within a shorter reporting deadline—meaning many manufacturers are facing a significant increase in effort during the validation process.
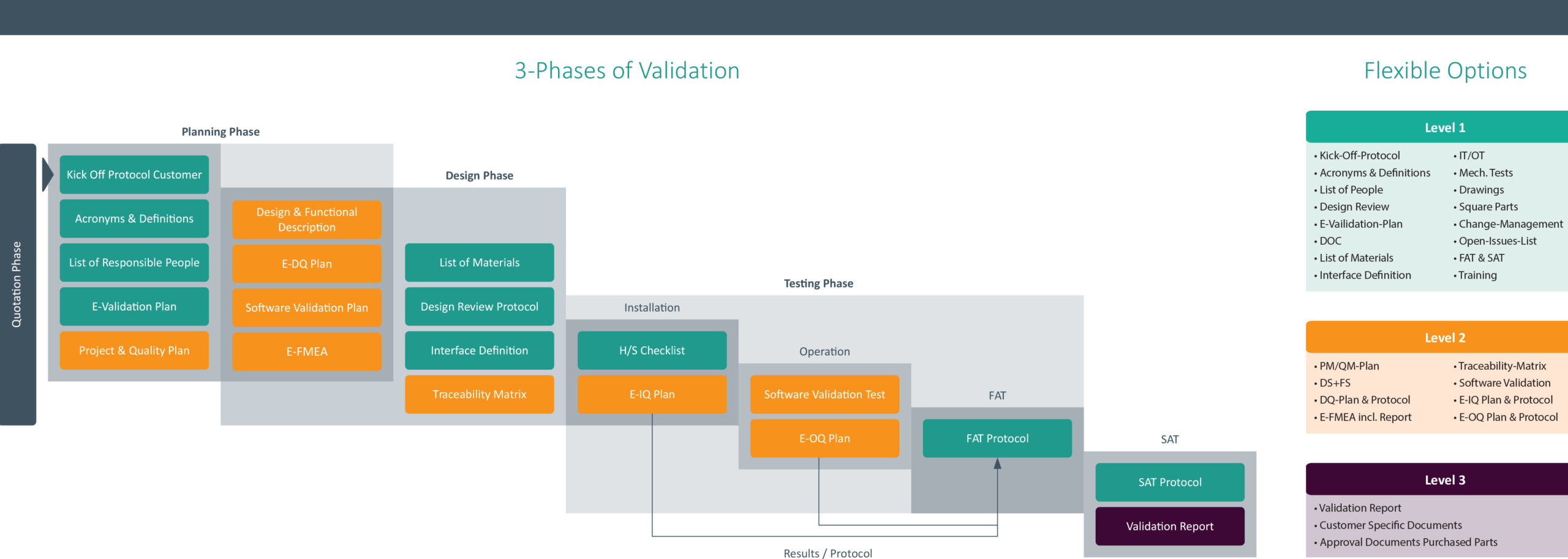
Invotec is here to help. Over the past six months, we have been working to develop a standard process that provides our customers with the best support. What we found is that by integrating our equipment validation into our project process, we can help ease the pressure of validation. Here’s how it works. At the beginning of the project, we will create a customized plan that supports the level of documentation you desire to satisfy the EU-MDR. Once the plan is in place, our team works with you throughout the project to ensure you have the necessary equipment information to meet your needs.
Below is a quick overview of our Three-Phase Validation Process along with the level of documentation you can choose from. Complete program information can be found here. This program is currently available at our European facility in Villingen-Schwenningen, Germany. We hope to roll out a similar program for US-based customers later this year.
Share this Post
